
Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. Molar Mass, Molecular Weight and Elemental Composition Calculator Enter a chemical formula to calculate its molar mass and elemental composition: Molar mass of HCl is 36. Since the molar mass of hydrogen chloride is about twice that of ammonia. One mole of 'something' contains 6.022 x 1023 entities. Engineering units use Btu and lb m for energy and mass units and psia for. Calculation procedure Molar mass of Hydrogen chloride, 36.
A mole is the unit that measures the amount of a substance. The mass of the earths atmosphere is estimated as 5.14 x 1015 t (1 t 1000 kg). The molarity of HCl is 11.65 M find the number of moles in 0.70 mL. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. Access the answers to hundreds of Molar mass questions that are explained in a way thats easy for you to understand. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms.
#Molar mass of hcl how to#
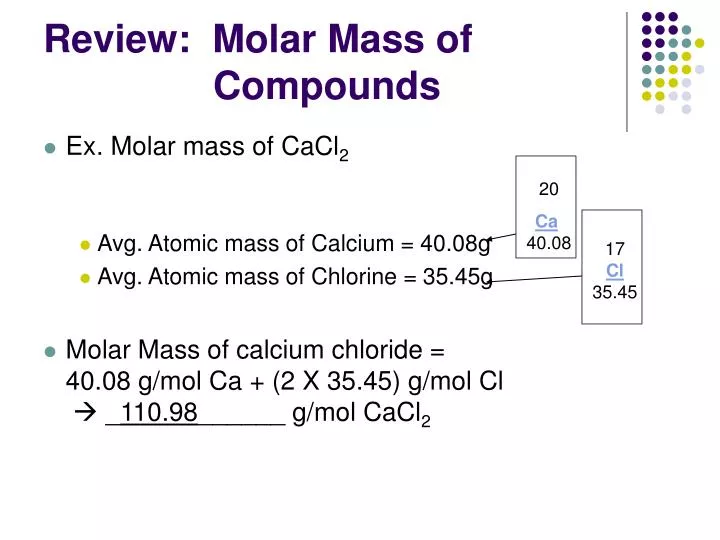
The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. One mole of a compound contains Avogadros number (6.022 x 1023) of molecules (molecular compound) or formula units (ionic compound).
